- 3. Results and discussion
Fig. 1 shows the effect of aluminum contents on the impact toughness at -20°C of the simulated CGHAZ as a function of (a) peak temperatures and (b) cooling time. As shown in
Fig. 1a,
Fig. 1 the impact toughness was gradually increased as aluminum contents increase from 0.02 to 0.3wt%. The drastic decrease of impact toughness with increase of the peak temperature above 1350°C was not observed with the aluminum contents above 0.3wt%. The impact toughness was slightly decreased with higher aluminum content of 0.45wt%. When aluminum content was increased above 0.6wt%, impact toughness was dramatically decreased at overall peak temperature. In the same manner, when the cooling time from 800 to 500°C was changed from 80 to 250 seconds with the fixed peak temperature of 1350°C (
Fig. 1b), the impact toughness was highly maintained with the aluminum content of 0.1 to 0.45wt%. The toughness of 0.02 and 0.06wt% aluminum added material was gradually decreased with increase of cooling time while 0.6 and 0.8wt% aluminum added materials show the extremely low impact toughness throughout the entire range of cooling time. As a result of the charpy impact tests, it could be concluded that impact toughness of simulated CGHAZ was increased with the appropriate aluminum contents in the range of 0.1 to 0.45wt%.
Fig. 1
Impact toughness of simulated HAZ of various Al added steels as a function of (a) peak temperature, (b) Δt 800-500
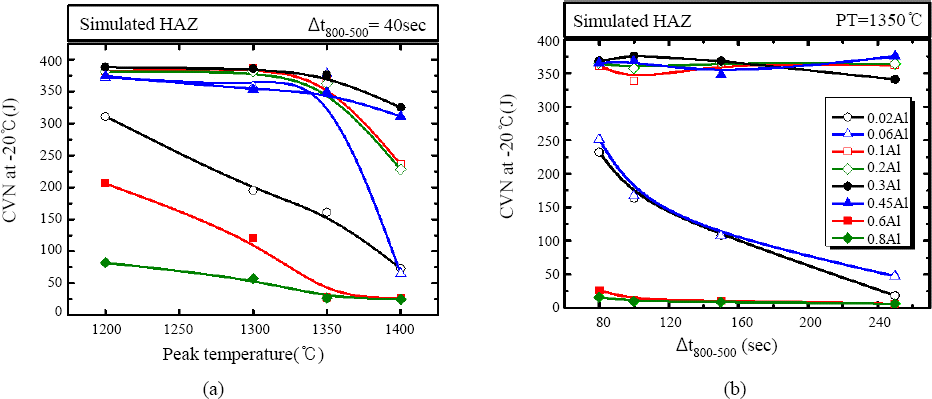
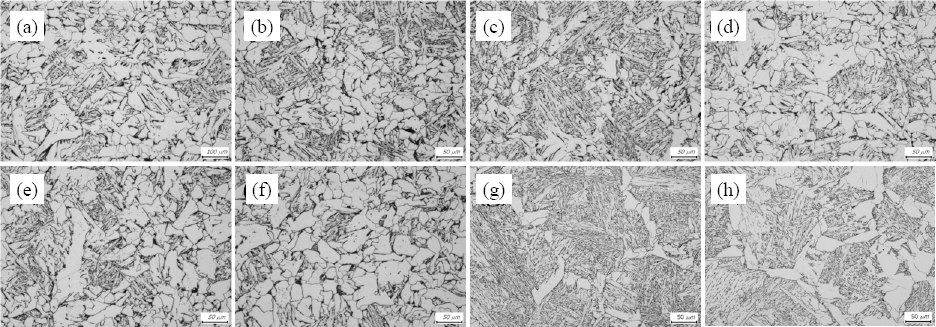
Fig. 2 shows the CGHAZ microstructures simulated with the peak temperature of 1400°C and cooling time from 800 to 500°C of 40 seconds. The simulated microstructures with the different aluminum content were primarily consist of bainite and ferrite. The fraction of pro eutectoid grain boundary ferrite was increased while the fraction of bainite was decreased as aluminum content increases from 0.02wt% up to 0.45wt%, as shown in
Fig. 2a through 2f. It could be concluded that the ferrite transformation is promoted by the accelerated ferrite nucleation as a result of the addition of ferrite stabilizer, aluminum
7,8). The increase of the start temperature of ferrite transformation with higher aluminum content was also observed by a series of additional dilatation test. However, the ferrite fractions in microstructures with higher aluminum content of 0.6 and 0.8wt% were remarkably decreased while the fraction of bainite was increased as shown in Figs.
2g and
2h.
Fig. 2
Microstructures of simulated heat affected zone (peak temp:1400°C, Δt 800-500=40sec) with different Al contents(wt%), (a) 0.02, (b) 0.06, (c) 0.1, (d) 0.2, (e) 0.3, (f) 0.45, (g) 0.6, (h) 0.8

The CGHAZs simulated with the peak temperature of 1350°C and cooling time from 800 to 500°C of 100 seconds (
Fig. 3) show the similar manner in microstructural changes as aluminum contents increase compare to the case of shorter cooling time, 40 seconds (
Fig. 2). The ferrite fraction increases and bainite fraction decreases as Al content increases up to 0.45wt% (
Fig. 3a through
3f). The size and fraction of ferrite in
Fig. 3 was bigger than
Fig. 2 due to the extended cooling time that allows the increase of transformation start temperature, nucleation, and growth of the ferrite. When the Al content were increased over 0.6wt% (Figs.
3g,
3f), the ferrite fraction was decreased and bainite fraction was increased remarkably, similar to Figs.
2g and
2h.
Fig. 3
Microstructures of simulated heat affected zone (peak temp:1350°C, Δt 800-500=100sec) with different Al contents(wt%), (a) 0.02, (b) 0.06, (c) 0.1, (d) 0.2, (e) 0.3, (f) 0.45, (g) 0.6, (h) 0.8
Palizdar
7) reported the effect of Al addition on the microstructure of 0.02C-0.3Si-1.4Mn-0.001N-(0.02~0.9) Al steels and observed the grain size refinement when the Al content was increased above 0.48wt%. He pointed out that there are two reasons for grain refinement with the Al contents above 0.48wt% as follows. The first is the pinning effect and austenite grain growth retardation by AlN precipitates that allow the ferrite grain refinement. However, this cannot be completely applicable in the present study due to the different kind of primary precipitates involved in the Ti-added steels used in this study.
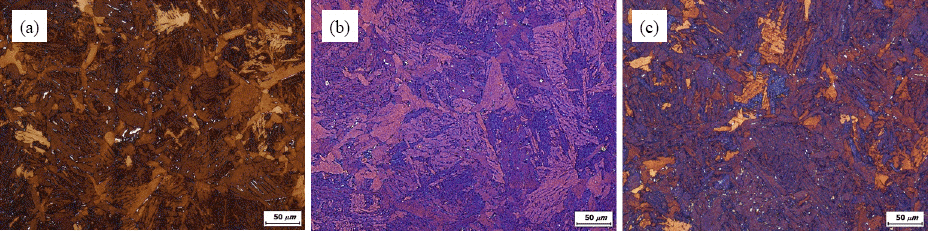
Fig. 4 shows the typical TEM micrograph from a carbon extraction replica and an energy probe x-ray micro analysis (EPMA) for a precipitate in the CGHAZ of 0.6Al added steel that was simulated with the peak temperature of 1400°C and 40seconds of the cooling time from 800 to 500°C. It has been revealed that the primary precipitates were TiN, and the secondary AlN and CuS were attached to the primary precipitate. The precipitation temperatures of TiN and AlN were above 1420°C, and near at 1100°C, respectively as a result of thermodynamic calculation. Therefore, the formation mechanism of the precipitates in
Fig. 4 could be explained as an agglomeration of each precipitate which has different temperature of precipitation. In short, TiN is the primary precipitate which affects the refinement of austenite grain rather than AlN in the CGHAZ of the steels used in this study. The second potential explanation for grain refinement with Al addition is segregation of Al at ferrite/austenite interfaces that could allow the retardation of ferrite growth, resulting in fine grain size. As shown in Figs.
2g,
2h,
3g, and
3h, the ferrite fraction of the CGHAZ was dramatically decreased above the Al content of 0.45wt% similar to Palizdar’s results of grain refinement when the Al content was increased over 0.48wt% while the quantity of AlN was almost fixed. It could be concluded that the drastic decrease of toughness of the CGHAZ with high Al content of 0.6 and 0.8wt% was due to the decrease of ferrite and increase of brittle bainite caused by Al partitioning at ferrite/austenite interface.
Fig. 4
Typical precipitates in simulated heat affected zone of 0.6Al added steel

Mintz
6) reported that the quantity and size of carbide could be diminished by 0.2~0.9wt% Al addition due to lower diffusivity of carbon in (0.02, 0.1%C)-(10,40ppm N) steel resulting in increase of HAZ toughness.
Fig. 5 shows the TEM micrograph and carbon profile in the CGHAZ of 0.03 and 0.3wt% Al added steels simulated with the peak temperature of 1400°C and cooling time from 800 to 500°C of 40 seconds. The finer and less quantity of cementite were observed in the 0.3wt% Al added steel (1.3% of the volume fraction in
Fig. 5c) compare to the 0.03wt% Al added steel (0.3% of the volume fraction in
Fig. 5a).
Table 2 shows the results of extracted residue analysis of 0.03 and 03wt% Al added CGHAZ in
Fig. 5. The extraction ratio was decreased remarkably from 37 to 6.2% when the Al content was increased from 0.03 to 0.3wt%, while Al was almost remained as solid solution in matrix. The quantity of Fe in residue was decreased from 2.95 to 2.23% as Al was increased from 0.03 to 0.3wt%. If the Fe contained precipitate is assumed to one hundred percent of cementite, the carbon content in cementite can be decreased from 0.21 to 0.16%. Therefore, it could be concluded that Al suppress the carbon diffusivity resulting in decrease of cementite precipitation.
Fig. 5
Carbide distribution in simulated heat affected zone. (a) and (b)TEM micrograph and carbon profile in 0.03%Al containing steel, respectively. (c) and (d) TEM micrograph and carbon profile in 0.3%Al containing steel, respectively

Table 2
Results of extraction residue analysis of simulated heat affected zone
|
Al(%) in matrix |
Al(%) in residue |
Extraction ratio(%) |
Fe(%) in residue |
C(%) in Fe3C |
|
0.03Al |
0.026 |
0.01 |
37 |
2.95 |
0.21 |
|
0.3Al |
0.3 |
0.002 |
6.2 |
2.23 |
0.16 |
Meanwhile, high free or solid solutioned aluminum in steel has been found to enhance the formation of M-A constituent
9). In the viewpoint of cementite precipitation, the effect of Al on preventing diffusion of carbon is advantage, but it could encourage the formation of M-A constituent, one of the most harmful element to toughness. M-A constituent is formed from carbon enriched austenite by the retardation of the transformation from austenite to ferrite and cementite during cooling
10) due to an excess of Al partition in proeutectoid ferrite and bainite. Figs.
6a,
6b,
6c show the M-A constituent distribution in the CGHAZ simulated with the peak temperature of 1400°C and 40seconds of the cooling time from 800 to 500°C. The volume fractions of M-A constituents were 0.63, 0.70, and 1.03% for 0.02wt% (
Fig. 6a), 0.1wt% (
Fig. 6b), and 0.3wt% (Fg.6c) of Al, respectively. The more Al contents, the higher area fraction of M-A constituent was observed, but it is important to note that the area fraction of M-A constituent was not too high below 2.0% and no massive type constituent was observed throughout all of the specimens in the condition of same CGHAZ simulation schedule.
Fig. 6
M-A constituents distribution of simulated heat affected zone (peak temp:1400°C, Δt 800-500=40sec) with different Al contents(wt%), (a) 0.02, (b) 0.1, (c) 0.3
Figs.
7a and
7b show the effect of nitrogen content on the impact toughness at -20°C of the simulated CGHAZ as a function of (a) peak temperatures and (b) cooling time from 800 to 500°C. 5, 41, and 100 ppm of nitrogen were added to 0.3wt% Al steels (No. 9, 5, 10 specimens in
Table 1). The impact toughness of the 5 ppm N added steel was gradually decreased as a peak temperature was increased while the toughness of 41 and 100ppm nitrogen added steel was maintained above 250J (
Fig. 7a). In the same manner, the impact toughness of the 5 ppm N added steel was gradually decreased as a cooling time was increased as shown in
Fig. 7b. The 41 ppm of nitrogen added steel shows the highest impact value throughout all over the test condition while 5 ppm of nitrogen added steel shows the lowest. Impact toughness of 100 ppm of nitrogen added steel was lower than that of the 41 ppm of nitrogen added steel, and the gap of value was about 100J. The difference of the impact toughness value as a function of the N content could be explained by the stability of TiN at high temperature and following austenite grain size near to the fusion line. The Ti to N ratio of 5, 41, 100ppm of nitrogen added steels are 36, 4.14, and 1.7, respectively. Therefore, it could be assumed that the thermal stability of the TiN is in following order, 100, 41, and 5 ppm of nitrogen added steels in the condition of almost same Ti content
11). As shown in
Fig. 8, the more N content, the more proeutectoid ferrite was formed while bainite fraction was decreased. The more ferrite was formed as cooling time increased from 40 to 100 seconds in the temperature range of 800 to 500°C in comparison with Figs.
8a through
8c and
8d through
8f. In case of the lowest, 5ppm of nitrogen added specimen, bainite fraction was highest due to the largest prior austenite grain size as shown in Figs.
8a,
8d. In addition, it could be concluded that lower toughness of 100 ppm nitrogen added steel than 41 ppm added steel was due to the excessive ferrite fraction and free N.
Fig. 7
Impact toughness of simulated HAZ with the different nitrogen contents, 5, 41, 100ppm as a function of (a) peak temperature, (b) Δt 800-500
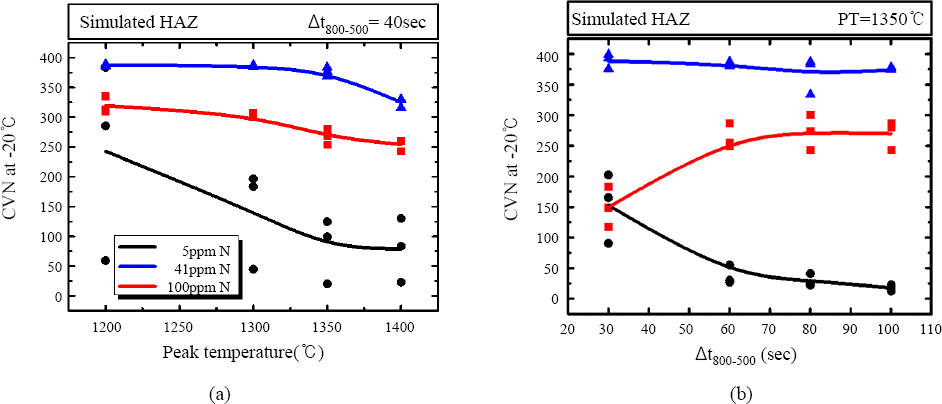
Fig. 8
Microstructures of simulated heat affected zone, (a), (b), (c) peak temp:1400°C, Δt 800-500=40sec, (d), (e), (f) peak temp:1350°C, Δt 800-500=100sec with the different nitrogen contents, 5ppm for (a), (d), 41ppm for (b), (e), 100ppm for (c) and (f)

Figs.
9a and
9b show the effect of 0.1 and 0.3wt% of silicon contents on the impact toughness of the simulated CGHAZ of 0.3wt%Al added steels as a function of (a) peak temperatures and (b) cooling time from 800 to 500°C (No. 11, 12 specimens in
Table 1). As shown in the Figs, the impact toughness of 0.3wt% Si added steels were lower than 0.1wt% Si added steels in the entire range of peak temperature and cooling time.
Fig. 10 shows the microstructure of simulated HAZ as a function of Si content and the peak temperature with the cooling time from 800 to 500°C of 40seconds for (a) 0.1wt%, 1350°C, (b) 0.3wt%, 1350°C, (c) 0.1wt%,1400°C, (d) 0.3wt%, 1400°C of Si content and the peak temperature, respectively. The less ferrite and more bainite were observed in 0.3wt% compare to 0.1wt% of Si added steel due to higher carbon equivalent. Besides the higher bainite fraction, the fraction of M-A constituent was another important factor which could determine the lower impact toughness of 0.3wt% of Si added steel CGHAZ. M-A constituent is promoted by Si addition due to the retardation effect of cementite precipitation resulting in carbon enrichment in austenite
9,12). As shown in
Fig. 11, more and bigger M-A constituent was observed in 0.3wt% of Si added steel (1.98% of the volume fraction in
Fig. 11a) compare to 0.1wt% of Si added steel (0.54% of the volume fraction in
Fig. 11b). In summary, it is important to determine the appropriate Si content concerning strength and toughness of the Al added steels.
Fig. 9
Impact toughness of simulated HAZ with 0.1 and 0.3wt%Si as a function of (a) peak temperature, (b) Δt 800-500
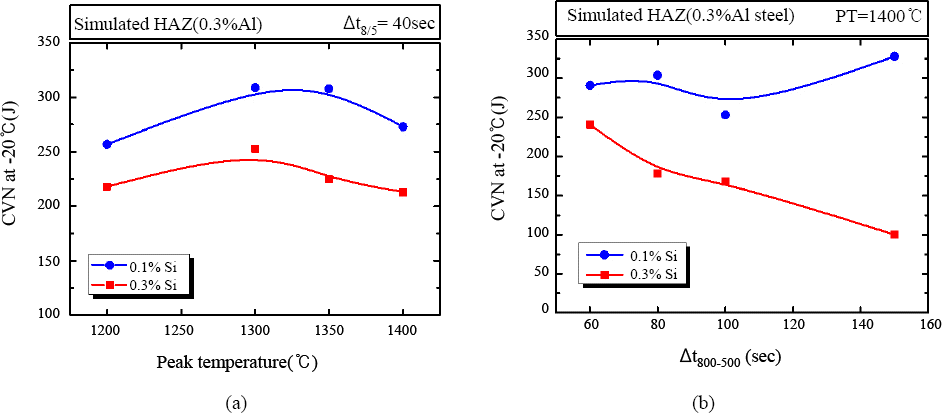
Fig. 10
Microstructures of simulated heat affected zone as a function of Si content and the peak temperature with the cooling time from 800 to 500°C of 40seconds for (a) 0.1wt%Si, peak temp.:1350°C, (b):0.3wt%Si, peak temp.:1350°C, (c) 0.1wt%Si, peak temp.:1400°C, (d) 0.3wt%Si, peak temp.: 1400°C

Fig. 11
M-A constituent in the simulated heat affected zone with the peak temperature of 1350°C and the cooling time of 40seconds from 800 to 500°C: (a) 0.3Al-0.3Si, (b) 0.3Al-0.1Si

 , Kyutae Han*
, Kyutae Han*
 Fig. 2 shows the CGHAZ microstructures simulated with the peak temperature of 1400°C and cooling time from 800 to 500°C of 40 seconds. The simulated microstructures with the different aluminum content were primarily consist of bainite and ferrite. The fraction of pro eutectoid grain boundary ferrite was increased while the fraction of bainite was decreased as aluminum content increases from 0.02wt% up to 0.45wt%, as shown in Fig. 2a through 2f. It could be concluded that the ferrite transformation is promoted by the accelerated ferrite nucleation as a result of the addition of ferrite stabilizer, aluminum7,8). The increase of the start temperature of ferrite transformation with higher aluminum content was also observed by a series of additional dilatation test. However, the ferrite fractions in microstructures with higher aluminum content of 0.6 and 0.8wt% were remarkably decreased while the fraction of bainite was increased as shown in Figs. 2g and 2h.
Fig. 2 shows the CGHAZ microstructures simulated with the peak temperature of 1400°C and cooling time from 800 to 500°C of 40 seconds. The simulated microstructures with the different aluminum content were primarily consist of bainite and ferrite. The fraction of pro eutectoid grain boundary ferrite was increased while the fraction of bainite was decreased as aluminum content increases from 0.02wt% up to 0.45wt%, as shown in Fig. 2a through 2f. It could be concluded that the ferrite transformation is promoted by the accelerated ferrite nucleation as a result of the addition of ferrite stabilizer, aluminum7,8). The increase of the start temperature of ferrite transformation with higher aluminum content was also observed by a series of additional dilatation test. However, the ferrite fractions in microstructures with higher aluminum content of 0.6 and 0.8wt% were remarkably decreased while the fraction of bainite was increased as shown in Figs. 2g and 2h. The CGHAZs simulated with the peak temperature of 1350°C and cooling time from 800 to 500°C of 100 seconds (Fig. 3) show the similar manner in microstructural changes as aluminum contents increase compare to the case of shorter cooling time, 40 seconds (Fig. 2). The ferrite fraction increases and bainite fraction decreases as Al content increases up to 0.45wt% (Fig. 3a through 3f). The size and fraction of ferrite in Fig. 3 was bigger than Fig. 2 due to the extended cooling time that allows the increase of transformation start temperature, nucleation, and growth of the ferrite. When the Al content were increased over 0.6wt% (Figs. 3g, 3f), the ferrite fraction was decreased and bainite fraction was increased remarkably, similar to Figs. 2g and 2h.
The CGHAZs simulated with the peak temperature of 1350°C and cooling time from 800 to 500°C of 100 seconds (Fig. 3) show the similar manner in microstructural changes as aluminum contents increase compare to the case of shorter cooling time, 40 seconds (Fig. 2). The ferrite fraction increases and bainite fraction decreases as Al content increases up to 0.45wt% (Fig. 3a through 3f). The size and fraction of ferrite in Fig. 3 was bigger than Fig. 2 due to the extended cooling time that allows the increase of transformation start temperature, nucleation, and growth of the ferrite. When the Al content were increased over 0.6wt% (Figs. 3g, 3f), the ferrite fraction was decreased and bainite fraction was increased remarkably, similar to Figs. 2g and 2h. Palizdar7) reported the effect of Al addition on the microstructure of 0.02C-0.3Si-1.4Mn-0.001N-(0.02~0.9) Al steels and observed the grain size refinement when the Al content was increased above 0.48wt%. He pointed out that there are two reasons for grain refinement with the Al contents above 0.48wt% as follows. The first is the pinning effect and austenite grain growth retardation by AlN precipitates that allow the ferrite grain refinement. However, this cannot be completely applicable in the present study due to the different kind of primary precipitates involved in the Ti-added steels used in this study. Fig. 4 shows the typical TEM micrograph from a carbon extraction replica and an energy probe x-ray micro analysis (EPMA) for a precipitate in the CGHAZ of 0.6Al added steel that was simulated with the peak temperature of 1400°C and 40seconds of the cooling time from 800 to 500°C. It has been revealed that the primary precipitates were TiN, and the secondary AlN and CuS were attached to the primary precipitate. The precipitation temperatures of TiN and AlN were above 1420°C, and near at 1100°C, respectively as a result of thermodynamic calculation. Therefore, the formation mechanism of the precipitates in Fig. 4 could be explained as an agglomeration of each precipitate which has different temperature of precipitation. In short, TiN is the primary precipitate which affects the refinement of austenite grain rather than AlN in the CGHAZ of the steels used in this study. The second potential explanation for grain refinement with Al addition is segregation of Al at ferrite/austenite interfaces that could allow the retardation of ferrite growth, resulting in fine grain size. As shown in Figs. 2g, 2h, 3g, and 3h, the ferrite fraction of the CGHAZ was dramatically decreased above the Al content of 0.45wt% similar to Palizdar’s results of grain refinement when the Al content was increased over 0.48wt% while the quantity of AlN was almost fixed. It could be concluded that the drastic decrease of toughness of the CGHAZ with high Al content of 0.6 and 0.8wt% was due to the decrease of ferrite and increase of brittle bainite caused by Al partitioning at ferrite/austenite interface.Mintz6) reported that the quantity and size of carbide could be diminished by 0.2~0.9wt% Al addition due to lower diffusivity of carbon in (0.02, 0.1%C)-(10,40ppm N) steel resulting in increase of HAZ toughness. Fig. 5 shows the TEM micrograph and carbon profile in the CGHAZ of 0.03 and 0.3wt% Al added steels simulated with the peak temperature of 1400°C and cooling time from 800 to 500°C of 40 seconds. The finer and less quantity of cementite were observed in the 0.3wt% Al added steel (1.3% of the volume fraction in Fig. 5c) compare to the 0.03wt% Al added steel (0.3% of the volume fraction in Fig. 5a). Table 2 shows the results of extracted residue analysis of 0.03 and 03wt% Al added CGHAZ in Fig. 5. The extraction ratio was decreased remarkably from 37 to 6.2% when the Al content was increased from 0.03 to 0.3wt%, while Al was almost remained as solid solution in matrix. The quantity of Fe in residue was decreased from 2.95 to 2.23% as Al was increased from 0.03 to 0.3wt%. If the Fe contained precipitate is assumed to one hundred percent of cementite, the carbon content in cementite can be decreased from 0.21 to 0.16%. Therefore, it could be concluded that Al suppress the carbon diffusivity resulting in decrease of cementite precipitation.
Palizdar7) reported the effect of Al addition on the microstructure of 0.02C-0.3Si-1.4Mn-0.001N-(0.02~0.9) Al steels and observed the grain size refinement when the Al content was increased above 0.48wt%. He pointed out that there are two reasons for grain refinement with the Al contents above 0.48wt% as follows. The first is the pinning effect and austenite grain growth retardation by AlN precipitates that allow the ferrite grain refinement. However, this cannot be completely applicable in the present study due to the different kind of primary precipitates involved in the Ti-added steels used in this study. Fig. 4 shows the typical TEM micrograph from a carbon extraction replica and an energy probe x-ray micro analysis (EPMA) for a precipitate in the CGHAZ of 0.6Al added steel that was simulated with the peak temperature of 1400°C and 40seconds of the cooling time from 800 to 500°C. It has been revealed that the primary precipitates were TiN, and the secondary AlN and CuS were attached to the primary precipitate. The precipitation temperatures of TiN and AlN were above 1420°C, and near at 1100°C, respectively as a result of thermodynamic calculation. Therefore, the formation mechanism of the precipitates in Fig. 4 could be explained as an agglomeration of each precipitate which has different temperature of precipitation. In short, TiN is the primary precipitate which affects the refinement of austenite grain rather than AlN in the CGHAZ of the steels used in this study. The second potential explanation for grain refinement with Al addition is segregation of Al at ferrite/austenite interfaces that could allow the retardation of ferrite growth, resulting in fine grain size. As shown in Figs. 2g, 2h, 3g, and 3h, the ferrite fraction of the CGHAZ was dramatically decreased above the Al content of 0.45wt% similar to Palizdar’s results of grain refinement when the Al content was increased over 0.48wt% while the quantity of AlN was almost fixed. It could be concluded that the drastic decrease of toughness of the CGHAZ with high Al content of 0.6 and 0.8wt% was due to the decrease of ferrite and increase of brittle bainite caused by Al partitioning at ferrite/austenite interface.Mintz6) reported that the quantity and size of carbide could be diminished by 0.2~0.9wt% Al addition due to lower diffusivity of carbon in (0.02, 0.1%C)-(10,40ppm N) steel resulting in increase of HAZ toughness. Fig. 5 shows the TEM micrograph and carbon profile in the CGHAZ of 0.03 and 0.3wt% Al added steels simulated with the peak temperature of 1400°C and cooling time from 800 to 500°C of 40 seconds. The finer and less quantity of cementite were observed in the 0.3wt% Al added steel (1.3% of the volume fraction in Fig. 5c) compare to the 0.03wt% Al added steel (0.3% of the volume fraction in Fig. 5a). Table 2 shows the results of extracted residue analysis of 0.03 and 03wt% Al added CGHAZ in Fig. 5. The extraction ratio was decreased remarkably from 37 to 6.2% when the Al content was increased from 0.03 to 0.3wt%, while Al was almost remained as solid solution in matrix. The quantity of Fe in residue was decreased from 2.95 to 2.23% as Al was increased from 0.03 to 0.3wt%. If the Fe contained precipitate is assumed to one hundred percent of cementite, the carbon content in cementite can be decreased from 0.21 to 0.16%. Therefore, it could be concluded that Al suppress the carbon diffusivity resulting in decrease of cementite precipitation.
 Figs. 7a and 7b show the effect of nitrogen content on the impact toughness at -20°C of the simulated CGHAZ as a function of (a) peak temperatures and (b) cooling time from 800 to 500°C. 5, 41, and 100 ppm of nitrogen were added to 0.3wt% Al steels (No. 9, 5, 10 specimens in Table 1). The impact toughness of the 5 ppm N added steel was gradually decreased as a peak temperature was increased while the toughness of 41 and 100ppm nitrogen added steel was maintained above 250J (Fig. 7a). In the same manner, the impact toughness of the 5 ppm N added steel was gradually decreased as a cooling time was increased as shown in Fig. 7b. The 41 ppm of nitrogen added steel shows the highest impact value throughout all over the test condition while 5 ppm of nitrogen added steel shows the lowest. Impact toughness of 100 ppm of nitrogen added steel was lower than that of the 41 ppm of nitrogen added steel, and the gap of value was about 100J. The difference of the impact toughness value as a function of the N content could be explained by the stability of TiN at high temperature and following austenite grain size near to the fusion line. The Ti to N ratio of 5, 41, 100ppm of nitrogen added steels are 36, 4.14, and 1.7, respectively. Therefore, it could be assumed that the thermal stability of the TiN is in following order, 100, 41, and 5 ppm of nitrogen added steels in the condition of almost same Ti content11). As shown in Fig. 8, the more N content, the more proeutectoid ferrite was formed while bainite fraction was decreased. The more ferrite was formed as cooling time increased from 40 to 100 seconds in the temperature range of 800 to 500°C in comparison with Figs. 8a through 8c and 8d through 8f. In case of the lowest, 5ppm of nitrogen added specimen, bainite fraction was highest due to the largest prior austenite grain size as shown in Figs. 8a, 8d. In addition, it could be concluded that lower toughness of 100 ppm nitrogen added steel than 41 ppm added steel was due to the excessive ferrite fraction and free N.
Figs. 7a and 7b show the effect of nitrogen content on the impact toughness at -20°C of the simulated CGHAZ as a function of (a) peak temperatures and (b) cooling time from 800 to 500°C. 5, 41, and 100 ppm of nitrogen were added to 0.3wt% Al steels (No. 9, 5, 10 specimens in Table 1). The impact toughness of the 5 ppm N added steel was gradually decreased as a peak temperature was increased while the toughness of 41 and 100ppm nitrogen added steel was maintained above 250J (Fig. 7a). In the same manner, the impact toughness of the 5 ppm N added steel was gradually decreased as a cooling time was increased as shown in Fig. 7b. The 41 ppm of nitrogen added steel shows the highest impact value throughout all over the test condition while 5 ppm of nitrogen added steel shows the lowest. Impact toughness of 100 ppm of nitrogen added steel was lower than that of the 41 ppm of nitrogen added steel, and the gap of value was about 100J. The difference of the impact toughness value as a function of the N content could be explained by the stability of TiN at high temperature and following austenite grain size near to the fusion line. The Ti to N ratio of 5, 41, 100ppm of nitrogen added steels are 36, 4.14, and 1.7, respectively. Therefore, it could be assumed that the thermal stability of the TiN is in following order, 100, 41, and 5 ppm of nitrogen added steels in the condition of almost same Ti content11). As shown in Fig. 8, the more N content, the more proeutectoid ferrite was formed while bainite fraction was decreased. The more ferrite was formed as cooling time increased from 40 to 100 seconds in the temperature range of 800 to 500°C in comparison with Figs. 8a through 8c and 8d through 8f. In case of the lowest, 5ppm of nitrogen added specimen, bainite fraction was highest due to the largest prior austenite grain size as shown in Figs. 8a, 8d. In addition, it could be concluded that lower toughness of 100 ppm nitrogen added steel than 41 ppm added steel was due to the excessive ferrite fraction and free N.
 Figs. 9a and 9b show the effect of 0.1 and 0.3wt% of silicon contents on the impact toughness of the simulated CGHAZ of 0.3wt%Al added steels as a function of (a) peak temperatures and (b) cooling time from 800 to 500°C (No. 11, 12 specimens in Table 1). As shown in the Figs, the impact toughness of 0.3wt% Si added steels were lower than 0.1wt% Si added steels in the entire range of peak temperature and cooling time. Fig. 10 shows the microstructure of simulated HAZ as a function of Si content and the peak temperature with the cooling time from 800 to 500°C of 40seconds for (a) 0.1wt%, 1350°C, (b) 0.3wt%, 1350°C, (c) 0.1wt%,1400°C, (d) 0.3wt%, 1400°C of Si content and the peak temperature, respectively. The less ferrite and more bainite were observed in 0.3wt% compare to 0.1wt% of Si added steel due to higher carbon equivalent. Besides the higher bainite fraction, the fraction of M-A constituent was another important factor which could determine the lower impact toughness of 0.3wt% of Si added steel CGHAZ. M-A constituent is promoted by Si addition due to the retardation effect of cementite precipitation resulting in carbon enrichment in austenite9,12). As shown in Fig. 11, more and bigger M-A constituent was observed in 0.3wt% of Si added steel (1.98% of the volume fraction in Fig. 11a) compare to 0.1wt% of Si added steel (0.54% of the volume fraction in Fig. 11b). In summary, it is important to determine the appropriate Si content concerning strength and toughness of the Al added steels.
Figs. 9a and 9b show the effect of 0.1 and 0.3wt% of silicon contents on the impact toughness of the simulated CGHAZ of 0.3wt%Al added steels as a function of (a) peak temperatures and (b) cooling time from 800 to 500°C (No. 11, 12 specimens in Table 1). As shown in the Figs, the impact toughness of 0.3wt% Si added steels were lower than 0.1wt% Si added steels in the entire range of peak temperature and cooling time. Fig. 10 shows the microstructure of simulated HAZ as a function of Si content and the peak temperature with the cooling time from 800 to 500°C of 40seconds for (a) 0.1wt%, 1350°C, (b) 0.3wt%, 1350°C, (c) 0.1wt%,1400°C, (d) 0.3wt%, 1400°C of Si content and the peak temperature, respectively. The less ferrite and more bainite were observed in 0.3wt% compare to 0.1wt% of Si added steel due to higher carbon equivalent. Besides the higher bainite fraction, the fraction of M-A constituent was another important factor which could determine the lower impact toughness of 0.3wt% of Si added steel CGHAZ. M-A constituent is promoted by Si addition due to the retardation effect of cementite precipitation resulting in carbon enrichment in austenite9,12). As shown in Fig. 11, more and bigger M-A constituent was observed in 0.3wt% of Si added steel (1.98% of the volume fraction in Fig. 11a) compare to 0.1wt% of Si added steel (0.54% of the volume fraction in Fig. 11b). In summary, it is important to determine the appropriate Si content concerning strength and toughness of the Al added steels.



